Overview Research
Neural mechanisms underlying innate and adaptive behavior in Drosophila
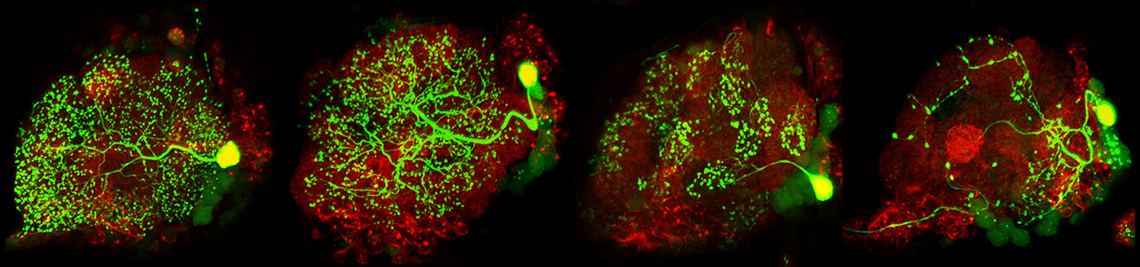
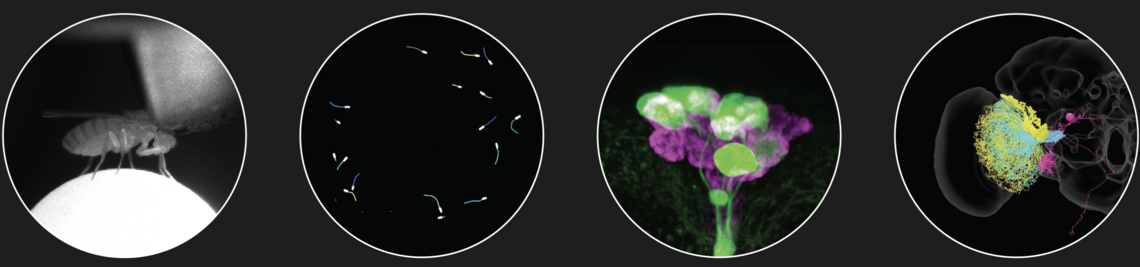
Our goal is to understand how sensory information is processed in the brain and how behaviour adapts to changing internal and external conditions. Using the fruit fly (Drosophila melanogaster) as a model organism, we focus our research on sensory processing, neuropeptide signalling, circadian biology and the neural control of behaviour. To this end, we employ advanced methods such as neurogenetics, in vivo two-photon microscopy, patch clamp, optogenetics and circuit reconstruction, as well as a wide range of behavioural assays for innate and learned behaviour.
To ensure survival in an ever-changing, complex world, animal behavior needs to be flexible and adaptive. Nervous systems have evolved to enable behavioral responses to a wide variety of sensory stimuli, but the adequate behavioral response to a given stimulus is highly context-dependent, and behavioral or internal states accordingly affect sensorimotor processing. For example, locomotion modulates responses of visual neurons, and hunger increases food-searching behavior and shifts taste preferences. Despite their ubiquitous importance, the neural mechanisms enabling context-dependent sensorimotor flexibility are not well understood. My Emmy Noether research program ‘Neural mechanisms enabling context-dependent sensorimotor flexibility’ aims to discover fundamental principles of motor control, in particular with regard to sensorimotor flexibility, by leveraging the power of neurogenetics, electron microscopy-based circuit reconstruction, and in-vivo patch-clamp recordings in behaving Drosophila.
Homepage Emmy-Noether-Programme
The Ache Lab is part of a NeuroNex Network with the goal of addressing the foundational question: How do biological nervous systems control and execute interactions with the environment?
Our network, which includes scientists and engineers from ten institutions across the United States, the United Kingdom and Germany, is focusing on Communication, Coordination, and Control in Neuromechanical Systems (C3NS) to develop comprehensive models of sensorimotor control with relationships to the environment, both within individual species, and across the phyla Arthropoda, Mollusca and Chordata.
Together, we seek to create a conceptual modeling framework that can predict control for organisms of different size and speed scales. Through our inter-phylum experimental study of sensorimotor control, we seek to identify convergent or conserved principles to refine and inform this framework. Such a framework will have a tremendous effect on the ability to interpret, and extend the impact of, experimental results across biology and robotics, with future applications to prosthetics.
The Ache Lab will closely collaborate with the Büschges, Ito and Blanke Labs at the University of Cologne and Nick Szczecinski’s Lab at the University of West Virginia to contribute a model of Drosophila motor control to the project. C3NS is led by Roger Quinn at Case Western Reserve University.
C3NS Homepage: https://case.edu/neuronex/
Objectives: The aim of the project is to characterise the contribution of growth-related neuroendocrine pathways to the effect of photoperiod on developmental time and body weight. Developmental time and final weight are important parameters for livestock production. In insects, developmental time and final body weight are tightly linked and are under control of two major neuroendocrine pathways: insulin- and prothoracicotropic hormone (PTTH) signalling. Signals from these pathways are integrated in the prothoracic gland (PG) and modulate the production of the key developmental hormone, the steroid ecdysone. In insects (including Drosophila), the photoperiod during development has a strong influence on developmental time and body weight. The underlying neuroendocrine and physiological mechanisms are, however, unclear.
Read more here: INCITE
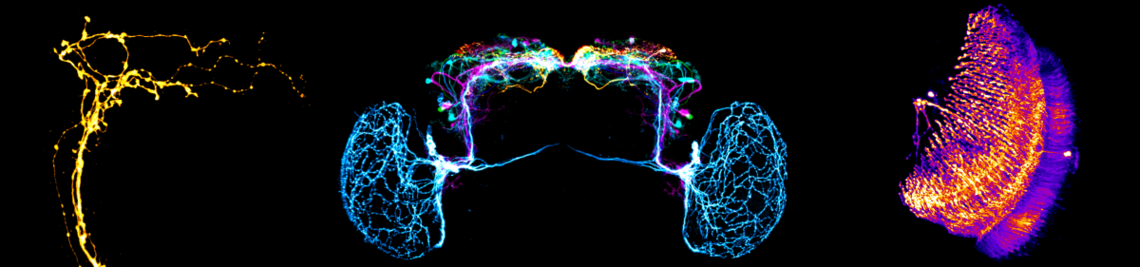
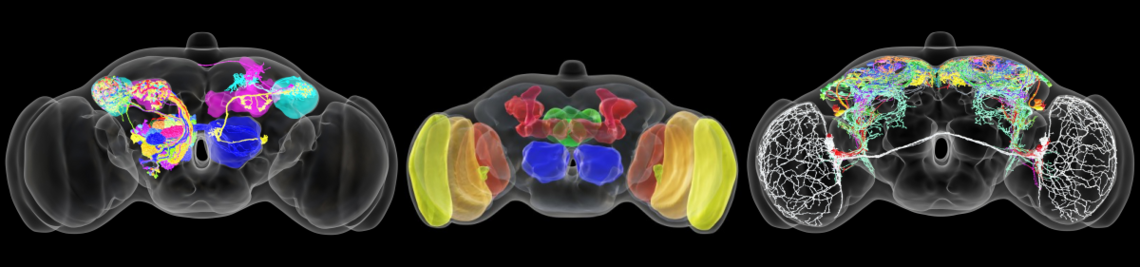
Inhibitory neurons are an essential component of nervous systems, balancing and modulating the output from excitatory neural subnetworks. Across olfactory systems in the animal kingdom, inhibition is particularly dominant. Highly complex and dense inhibitory circuits control the impact of sensory neuron input. Yet more strikingly, inhibitory neurons are the main mediators of interactions between second order principal neurons that lack direct synaptic contacts with each other. These pathways are thought to regulate olfactory sensitivity in a state-dependent manner, provide gain control, synchronize the spiking activity of principal neuron ensembles, and enhance the contrast of representations of similar odorants via decorrelation of their response patterns. The architecture of the underlying network anatomy is astonishingly similar across insects and vertebrates - a prime example of convergent evolution. Here we aim to further clarify the cellular correlates of defined inhibitory interactions within early olfactory processing across phylae. More specifically, we will investigate the cellular basis and functional impact of dominant forms of inhibition within the fly antennal lobe and the rat olfactory bulb, namely recurrent inhibition and isotropic and anisotropic lateral inhibition, the latter allowing for directed interactions between individual glomerular channels that might be even hard-wired. Beyond the first phase of the SPP funding period, that was focussed on anisotropic inhibition across phylae and in partly delayed due to the pandemic, we now plan to also identify the anatomical correlates of recurrent inhibition and isotropic lateral inhibition in the fly, to ultimately assign the mentioned specific inhibitory interactions to defined interneuron types. Next, we will test these assignments and their functional impact in experiments that involve targeted silencing of the respective interneuron subtypes during innate and learned odor-guided behavior. In parallel we plan to integrate the results of these and previous investigations into lineage-specific network models that are based on realistic neuroanatomical parameters derived from recent ultrastructural data and functional data from our own and others work. Ultimately, we aim to use these models to develop a new generic network model of the convergent olfactory system, in tight collaboration with other members of the priority program with computational and circuits expertise.
Animals have to adapt to seasonal changes in the environment. A too late adaptation to the coming winter will definitively cause their death. Insect start to hibernate (=diapause) when temperatures drop and day-length (photoperiod) decreases below a certain critical value. It is generally thought that the circadian clock is crucial to measure day-length, but the mechanisms how this is done and how the signals about day-lengths are transformed to the diapause inducing hormonal centres in the insect brain are largely unknown.
This Marie Curie Network aims to uncover the mechanisms of photoperiodic control in four model species: The parasitoid wasp, Nasonia vitripennis, the Lindenbug, Pyrrhochoris apterus, the Olive fly, Bactrocera oleae and the genetic model system Drosophila melanogaster. ..more
Funded at the Julius-Maximilians-University of Würzburg from 2013 to 2017
Timing plays an important role in all living systems. Endogenous clocks enable organisms to anticipate and adapt to daily or seasonal variation in environmental conditions. Being at the right time at the right place crucially determines the chance to find sufficient food, the success of mating, the success in raising brood, the chance to synchronise with mutualistic interaction partners and the likelihood to escape antagonists or harmful environmental events. Thus, proper timing of development, maturation, population phenology and a wide range of different behaviours is of paramount importance for the reproductive success and survival of all animals. Under laboratory conditions, the molecular basis of daily timing via endogenous clocks is intensively studied in few model organisms such as Drosophila melanogaster. However, little is known about the functional impact of such clocks in real nature. Our knowledge on timing mechanisms on time scales exceeding one day is even scarcer and restricted to few species. We also do not know how endogenous clocks and other timing mechanisms promote the adaptation to changing environmental conditions. Further, the impact of clocks and other evolutionary achieved timing mechanisms on the fitness of individuals, eusocial insect colonies, or on the synchronisation of multitrophic biotic interactions are little understood.
The Collaborative Research Centre (CRC) addresses these important questions using anintegrative approach. We focus on insects, since insects are in particular masters in behavioural timing and in adapting quickly to changing environmental conditions, which made them the predominant animal taxon on our planet with enormous ecological impact. We investigate mechanisms of proper timing in selected species at the molecular, cellular and neuronal networklevels, analyze the influence of timing on individual behaviour and interactions in social groups,populations, and communities, and, ultimately, determine the impact of proper timing on fitness. Each of these levels is important for its own sake. Yet, the new and important step we take here is to integrate the analyses of the different proximate/mechanistic levels of timing into an ecological context (success on a daily base, throughout the individual life span, and across generations).
The proposed CRC intends to establish a platform integrating different biological subdisciplines
ranging from molecular biology via neuroethology to ecology in order to promote vivid synergistic interactions among them. The research of this CRC goes beyond current studies in the individual biological disciplines and brings together ecological concepts with mechanistic research on the molecular, neuronal and organismic level. Our long term goal is to elucidate the proximate mechanisms underlying timing, and at the same time, unravel the ultimate consequences of proper timing for fitness in real ecosystems and in the context of global environmental change.