ME 4866/1-2
ME 4866/1-2: Evolutionary and seasonal adaptation of daily activity and of the circardian clock parameters in Drosophila
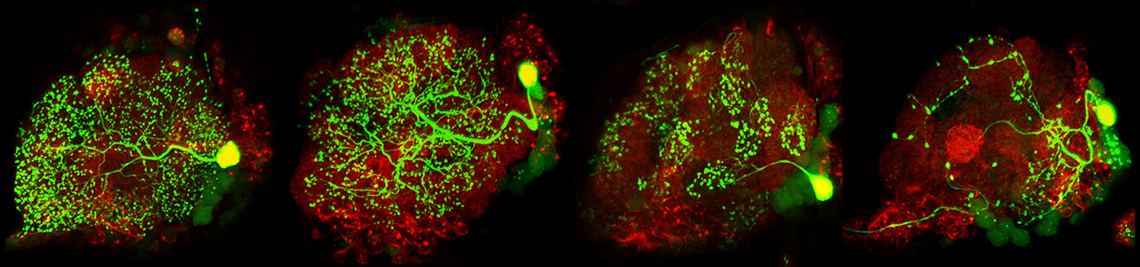
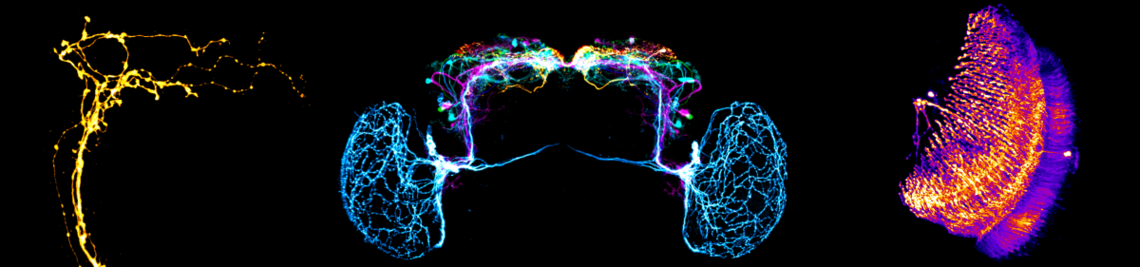
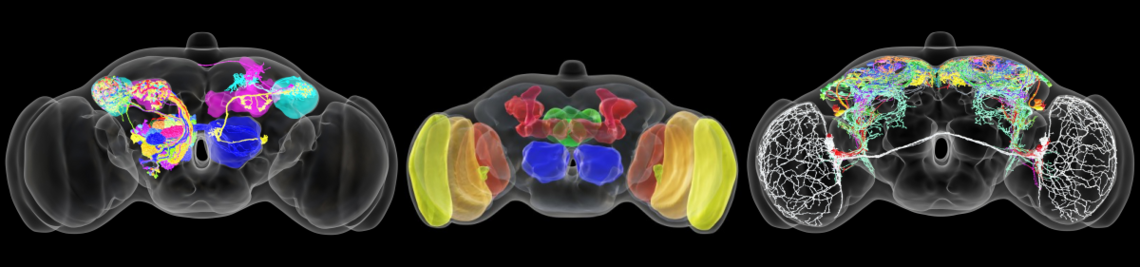
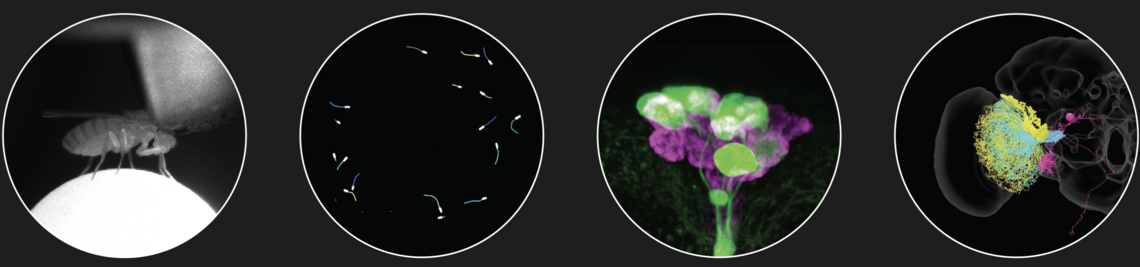
The family of Drosophilids include tropical, cosmopolitan and subarctic species. The aims of the present study are to identify the neuronal clock mechanisms that are crucial for the adaptation of daily activity to different latitudes (project 1) and the molecular clock mechanisms that allow measuring day length and preparing for the winter (project 2). In project 1, we identified the neuronal clock network in the brain of the originally tropical Drosophila melanogaster as the ancestral one and that there are different ways to adapt the circadian system to a life at high latitudes, which all have one thing in common: they make the circadian clock more plastic so that the daily activity can easily follow the changing photoperiods. One way to do so is to lose the neuropeptide PDF (Pigment-Dispersing Factor) in some clock neurons and the blue-light photopigment CRY (Cryptochrome) in others (e.g. in flies of the virilis group). Other ways are to eliminate the rhythmic secretion of PDF (e.g. in Chymomyza flies) or to reduce the light-sensitivity of CRY or its ability to interact with the clock protein TIM (Timeless) (e.g. in D. funebris). In the second funding period, we aim to finish the already started analysis on ~30 Drosophila species stemming from different latitudes in order to gain deeper insights how circadian clocks evolved and adapted to the environment. Furthermore, we will focus on the clock of D. funebris because this clock appears unusally well adapted to a life at temperate and arctic regions and promises new insights into the evolution of high-latitude clocks. In project 2, we found that the distance between the maxima of the clock proteins PER (Period) and TIM in the clock neurons of D. melanogaster codes for day length and modulates frost resistance: under short photoperiods the maxima of these cycling clock proteins are close together and the flies are more frost resistant as under long photoperiods, in which the PER and TIM maxima are far apart. Furthermore, we identified 79 candidate genes that change their expression in specific clock neurons under short and long photoperiods. Now, we plan to identify the most promising genes by comparison of their expression patterns in high-latitude Drosophila species and D. suzukii, then manipulate them in D. melanogaster to study the flies’ thermal tolerance. Furthermore, we aim to identify the specific clock neurons in the brain of D. melanogaster that are most important for seasonal adaptation of frost resistance. This will not only allow a better understanding of the precise role of D. melanogaster’s clock in seasonality but possibly also unravel how the clock modifications in high-latitude species evolved. We will also generate per and tim null mutants in D. suzukii using the CRISPR-Cas9 technique to reveal the role of the circadian clock for seasonality in a fly with strong morphological and behavioural changes between summer and winter. Altogether our studies promise to unravel the clock mechanisms leading to seasonal adaptation.