Böttcher Lab
Welcome to the Boettcher Group
Electron cryo Microscopy of Biological assemblies and Macromolecular complexes
The structural organization of molecular components determines the underlying mechanisms of biological processes. To understand these processes, we need to know
the structures of the molecular players involved. We use electron cryo microscopy to determine the structures of these components in different functional and conformational states. Our current research focuses on protein assemblies that participate in processes in, on, and across membranes.
We use a multidisciplinary experimental approach that combines structural, biochemical and biophysical information. At the heart of our methodological portfolio is structure determination by electron cryo microscopy and image processing. This is a powerful method for elucidating the structure and conformational plasticity of biological assemblies. The method allows imaging of complexes “in solution” under a wide range of conditions in different functional states.
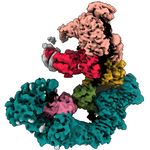
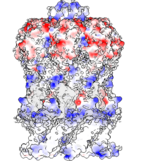
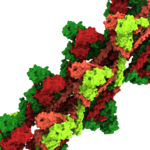
Currently, we investigate mechanosensitive channels of small conductance (MscS-like channels). These channels sense membrane tension and gate when the tension exceeds a certain threshold. We have found that different MscS-like channels follow different conformational routes in this process. We also investigate Lymphostatin, a huge virulence factor of pathogenic E.coli. Lymphostatin helps the bacteria to colonize the gut by enhancing bacterial attachment and reducing mitogen activated T-cell proliferation. Our structures reveal that Lymphostatin is a highly dynamic multi-domain protein with some domains resembling enzymes but lacking the necessary motifs to support activity.
Our future research on MscS-like channels will aim to identify the minimal channel architecture and reprogram the gating and sensing characteristics. For Lymphostatin, we will investigate how the different functionalities of the protein are released and how this compares to related virulence factors of other bacteria.
Website : Rudolf-Virchow-Zentrum