Protein kinase RSK and nervous system dysfunction
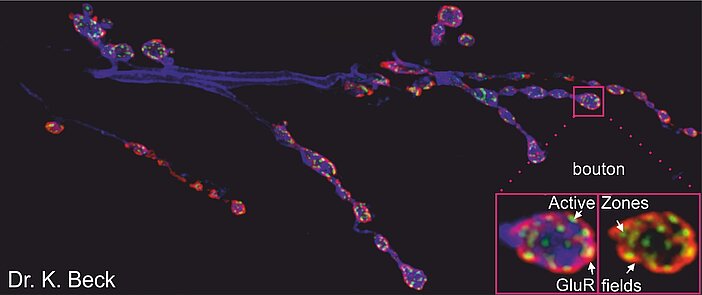
In this project, we are focusing on the Coffin-Lowry-syndrome (CLS) associated protein kinase RSK2. Accumulating evidence from different model organisms point to neurophysiological dysfunction as a main cause for the severe intellectual disabilities seen in CLS patients. Using the Drosophila motoneuron system as an easily accessible model, we could demonstrate an involvement of this kinase in synaptic transmission, axonal transport and regulation of intracellular ERK signaling. Furthermore, we uncovered a function of RSK in the Drosophila circadian clock to maintain the 24 hour activity rhythm. Our current aim is to study the role of RSK in central brain neurons in the context of olfactory learning and synaptic plasticity and to identify molecular targets.
Key publications
Fischer, M., Raabe, T. (2018) Animal models for Coffin-Lowry Syndrome: RSK2 and nervous system dysfunction. Front. Behav. Neurosci. 12, 106.
Beck, K., Hovhanyan, A., Menegazzi, P., Helfrich-Förster, C., Raabe, T. (2018) RSK influences the pace of the circadian clock by negative regulation of protein kinase Shaggy activity. Front. Mol. Neurosci. 11, 122.
Beck, K., Ehmann, N., Andlauer, T.F.M., Ljaschenko, D., Strecker, K., Fischer, M., Kittel, R.J., Raabe, T. (2015) Loss of the Coffin-Lowry syndrome-associated gene RSK2 alters ERK activity, synaptic function and axonal transport in Drosophila motoneurons. Dis. Models Mech. 8, 1389-1400.



