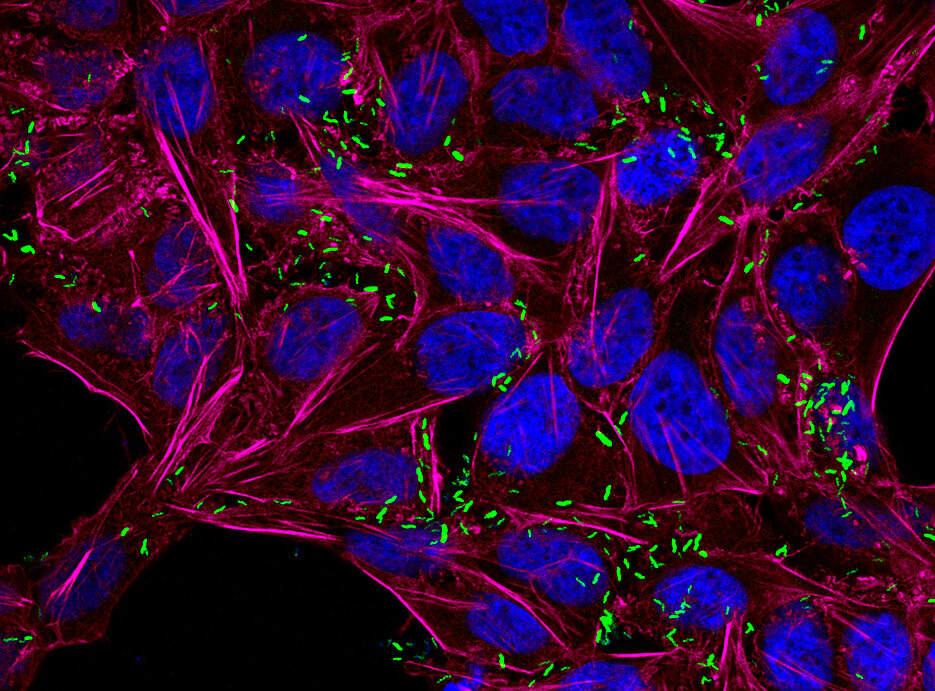
Fighting Against Multi-Resistant Bacteria
02/11/2020Many bacterial pathogens develop resistance to antibiotics. In their search for new therapeutic strategies, Würzburg research groups employ modern digital technologies. The Free State of Bavaria provides millions for this purpose.
Developing fundamentally new approaches against multi-resistant germs: This is the goal of the new Bavarian research network bayresq.net. The Free State of Bavaria is providing over ten million euros for this purpose. Research groups from Julius-Maximilians-Universität (JMU) Würzburg are involved in two of the six funded projects. They will receive around 2.7 million euros over the next five years.
Using digital methods for designing RNA antibiotics
Conventional antibiotics are usually effective against a broad spectrum of bacteria. As effective as they may be, they unfortunately also promote the development of multi-resistant germs. They also destroy the protective microbiota – including bacteria that have useful effects in the human intestine.
Using an interdisciplinary approach, a JMU team led by Professor Jörg Vogel, junior Professor Lars Barquist and junior research group leader Dr. Franziska Faber will investigate new antibiotics that can be used to treat specific types of bacteria. In principle, such novel antibiotics based on the nucleic acid RNA can be "programmed" according to simple chemical principles and can also be reprogrammed if resistance occurs.
"Digitalisation has arrived in the life sciences since quite a while ago and the problem of antibiotic resistance must be approached from different angles," says Jörg Vogel. "The funding provided by bayresq.net will allow us to develop digital strategies to combat multi-resistant germs that go hand in hand with our work in the laboratory".
The aim of the project at the JMU Institute of Molecular Infection Biology / Research Center for Infectious Diseases (IMIB / ZINF) is to develop universal rules for programming effective antibiotics and to decipher the underlying mechanisms. For this purpose, the team uses a combination of high-throughput sequencing and machine learning. The aim is to create a very powerful digital platform for the future application of RNA-based antibiotics against a variety of bacteria.
Salmonella and Campylobacter under pressure
Pathogens are exposed to diverse chemical stimuli and stress conditions during the infection process. These stimuli originate from the host organism, the microbiota and food, as well as from antibiotics and other drugs. Pathogens have various survival and adaptation strategies to adapt to these changing conditions. However the molecular mechanism of how a given stimuli activates particular adaptation responses is largely unknown.
This is where the StressRegNet consortium comes in, which is led by Professor Cynthia Sharma from tthe IMIB / ZINF and junior research group leader Dr. Ana Rita Brochado from the ZINF / Biocenter together with Professor Christian Müller from the Institute of Statistics of the Ludwig-Maximilians-Universität Munich.
The team will investigate different chemical stimuli and regulatory signaling pathways that control host adaptation of Salmonella and Campylobacter. Both are two widespread food-borne pathogens that have recently been classified by the World Health Organization (WHO) with high priority for research and development of new antibiotics.
Using high-throughput automation technologies, the researchers aim to expose the two bacterial pathogens to a library of more than 3,000 different small molecules. The gene expression responses and stress reactions triggered by these chemical molecules will then be measured, with a focus on regulation by small regulatory RNA molecules. The measurements will provide a highly complex dataset. This will then be investigated using machine learning techniques for specific signals and stress responses related to antibiotic sensitivity and host interactions.
The StressRegNet consortium aims to achieve a significant advance in automation and digitalization in infection biology. At the same time, its work should provide valuable new insights into the regulatory networks of bacteria. This is essential for the development of new antimicrobial strategies.
Facts about bayresq.net
The six projects of the bavarian research network "New strategies against multi-resistant pathogens using digital networking – bayresq.net" are running at JMU, the University of Erlangen-Nuremberg, LMU, the Technical University of Munich and the University of Regensburg. Research groups from biology, bioinformatics, chemistry, biophysics, medicine and mathematics are involved.
In addition to junior Professor Lars Barquist from JMU/HIRI, two junior research group leaders from the ZINF are represented in the Würzburg projects. Dr. Franziska Faber has been researching at ZINF since 2018; Dr. Ana Rita Brochado was appointed as a junior research group leader at the ZINF / Biocenter (Chair of Microbiology, Professor Thomas Rudel) in 2019. Her group is also supported by the Emmy-Noether-Programme of the German Research Foundation (DFG).
ZINF: Young Investigators Groups as the core
The Research Center for Infectious Diseases (ZINF) at JMU was founded in 1993 and is the oldest university institution in Germany dedicated to interdisciplinary and cross-faculty research on infectious diseases. Since 2010, it has been a central scientific institution of the JMU.
At the heart of ZINF are the independent junior research groups, which give young, talented researchers the opportunity to set up their own laboratory after their postdoctoral research. Over the past 25 years, the ZINF junior research groups have developed into an internationally renowned programme. The success of this program is also reflected in the fact that many former junior research group leaders have been appointed to national and international professorships or permanent group leader positions after their time at ZINF.
"The ZINF young investigator programme has enabled us to steadily recruit new expertise in emerging technologies or novel research areas in the field of infection biology to JMU over the last years . For example, Dr. Brochado's expertise in the field of genome-wide screens using high-throughput automation technologies is essential for our StressRegNet consortium," says Cynthia Sharma.
Web link
Bavarian research network bayresq.net
Contact persons
Prof. Dr. Jörg Vogel, Director of the Institute of Molecular Infection Biology, T +49 931 31-82575, joerg.vogel@uni-wuerzburg.de
Website: http://www.imib-wuerzburg.de
Prof. Dr. Cynthia Sharma, Spokesperson of the Research Center for Infectious Diseases (ZINF), +49 931 31-82560, cynthia.sharma@uni-wuerzburg.de
Website: https://www.uni-wuerzburg.de/zinf/