Group PD Dr. Susanne Berger
PD Dr. Susanne Berger
Lehrstuhl Pharmazeutische Biologie
Julius-von-Sachs-Institut für Biowissenschaften
Julius-von-Sachs-Platz 2
97082 Würzburg
Phone: + 49 931 31-86170
Fax: + 49 931 31-86182
susanne.berger@uni-wuerzburg.de
Research topics
Oxylipins are derived from fatty acids and function as signals to regulate growth and development as well as responses to stress. The role of jasmonic acid and its derivatives has been intensively studied in plants. In contrast, many questions regarding the significance and mechanisms of action of other oxylipins are still open. In functional approaches using mutants with defects in the formation or recognition of these signals, we are investigating the biological function of various oxylipins. One focus is the response to biotic and abiotic stress factors. Furthermore, we analyze the biological activities of oxylipins at the transcript and metabolite level as well as their effects on growth and photosynthesis. Another focus of our research is the elucidation of signaling mechanisms from recognition of oxylipins to the downstream responses such as regulation of gene expression. The thale cress (Arabidopsis thaliana) and the liverwort Marchantia polymorpha are used as model organisms.
Res-oxylipin signaling in Arabidopsis
Mechanisms of recognition and signal transduction have been described for jasmonic acid and its derivatives, but little is known about how the effects of reactive electrophilic oxylipins such as 12-oxo-phytodienoic acid are mediated. In order to identify components of these signaling pathways, mutants were isolated that show an altered response to oxylipins such as prostaglandins and thus presumably have a defect in a component of the signal transduction chain. Reporter plants expressing the luciferase gene under the control of an oxylipin-responsive promoter were used for the mutant screen. Using this approach, a zinc finger protein was identified as a novel factor of oxylipin signal transduction. To understand the relevance of this protein and of oxylipins in general in stress tolerance, the response to biotic and abiotic stress factors is tested in different mutants with altered expression of the zinc finger protein and related proteins. We are interested in the effects on the physiological level as well as differences in the expression of various oxylipin-responsive genes and the metabolite profile.
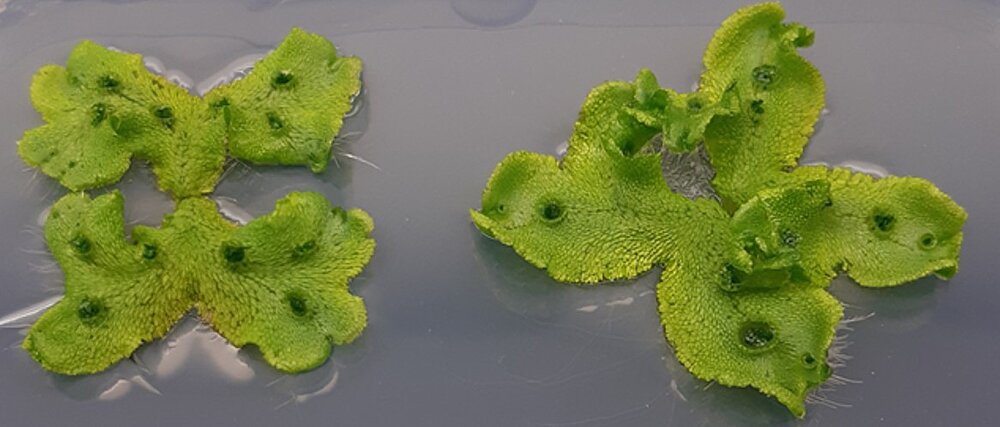
Oxylipin-signaling in liverwort
Oxylipins play an important signaling role in many organisms. While in vascular plants jasmonic acid and its derivatives regulate growth, development and stress responses, this oxylipin is missing in the liverwort Marchantia polymorpha. Here, the reactive electrophilic oxylipin dinor-oxo-phytodienoic acid takes over most of the functions of jasmonic acid. Despite differences in the oxylipin profile, some oxylipin signal transduction factors are conserved between Arabidopsis and Marchantia. Marchantia polymorpha has been established as a model system because it has a smaller genome than vascular plants and therefore fewer proteins are responsible for similar functions. To elucidate the relevance of the zinc finger protein in Marchantia polymorpha, mutants with defects in this protein will be generated using CRISPR/Cas technology and the effects on growth, development and stress tolerance will be investigated. The comparison with Arabidopsis also allows us to draw conclusions about the evolution of oxylipin signaling and signal transduction mechanisms.
Lipid oxidation upon biotic stress
Triacylglycerols (fat) are lipids with important energy storage function in seeds and fruits of plants. The content in vegetative tissue is low, but various abiotic stress factors and contact with fungal or bacterial pathogens also lead to an increase in the storage lipid content in the leaves and roots of plants. This increase can also be triggered by elicitor molecules originating from bacteria or fungi. Little is known about the signal transduction chains that mediate this reaction. Furthermore, treatment with pathogens or elicitors leads to oxidation of storage and membrane lipids. Oxidation can be catalyzed enzymatically or non-enzymatically by reactive oxygen species, in particular polyunsaturated fatty acids are oxidized. We investigate the mechanisms of lipid accumulation and lipid oxidation as well as signal transduction in roots and leaves of Arabidopsis after contact with bacteria, fungi and lipopolysaccharide, a bacterial elicitor.