Research Dröge-Laser Lab
Tpoic A: Energy Homeostasis
Topic B: Arabidopsis Transcription Factors
Plant energy management in response to energy limiting conditions
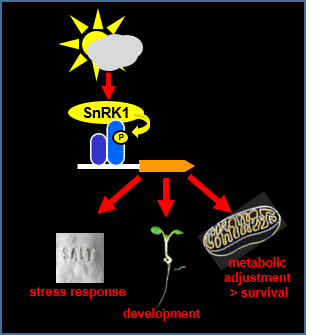
Sustaining energy homeostasis is crucial to every living being. To balance energy supply and demand, plants make use of an evolutionarily conserved managing system consisting of two counteracting kinases: TOR (TARGET OF RAPAMYCIN) supports anabolic, energy-consuming metabolism, whereas SnRK1 (Snf1-RELATED PROTEIN KINASE1) activates catabolic, energy-preserving responses. Natural conditions such as low-light, abiotic or biotic stress lead to energy depletion. Recently, we identified members of the bZIP (basic leucine ZIPPER) transcription factor family belonging to groups C and S1 as target proteins and SnRK1 downstream mediators. In this project, we (1) study the mechanism of transcriptional control, facilitated by SnRK1 and C/S1-bZIPs and (2) define the functional aspects they are regulating. Here, we analyse alternative pathways in amino acid catabolism to support mitochondrial ATP Production and survival upon energy deprivation, energy-depending developmental processes related to root architecture, seedling development, dark-induced and natural senescence, as well as salt stress and pathogen defense.
References: Pedrotti et al., Plant Cell 2018; Dröge-Laser and Weiste, TIPS 2018; Mair et al., eLife 2015, Hartmann et al., Plant Cell 2015, Nukarinen et al., Sci. Rep. 2016, Dietrich et al., Plant Cell 2011
Co-worker: Christoph Weiste, Jan Draken, Philipp Kreisz, Theresa Wildenhain
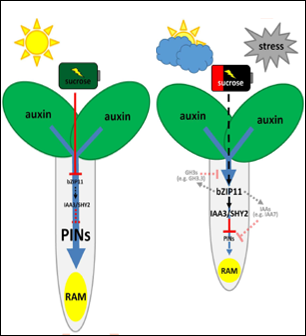
Fine-tuning of auxin-mediated growth according to the cellular energy resources
The plant hormone auxin controls various aspects of plant growth and development. Most of the auxin responses are regulated on a transcriptional level, facilitated by Auxin Response FACTORS (ARFs). Plant growth dependents on the available energy, C- and N-resources. Here, we follow the hypotheses that C/S1 bZIP integrate information on cellular energy reserves into auxin mediated growth responses.
References: Weiste et al., PLoS Genetics 2017; Weiste und Dröge-Laser, Nature Com. 2014
Co-worker: Christoph Weiste
Molecular tools to study the Arabidopsis Transcription factor ORFeome
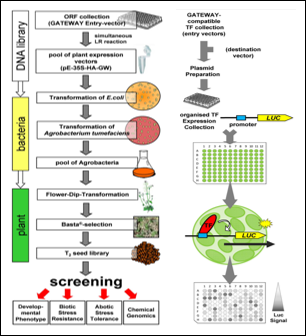
Transcription factors (TFs) are important cellular regulators. In the model plant Arabidopsis thaliana more than 2000 TF genes have been identified and cloning approaches generated large Open Reading Frame (ORF) collections. Nevertheless, tools are required to functionally characterise this “ORFeome”.
> Arabidopsis thaliana Transcription Factor Open Reading Frame Expression (AtTORF-Ex)
We have developed a batch plant transformation procedure to generate pooled collections of plants overexpressing TF genes (Arabidopsis thaliana TF ORF EXpression, AtTORF-EX), which can be used to define TF function in stress response and development.
> Protoplast Trans Activation (PTA) Screening
A high-throughput microtiter plate based Protoplast Trans-Activation (PTA) system has been established which can be used in screening approaches to define which TFa are regulating a given promoter in planta. A screening population of approximately 1700 GATEWAY compatible TF expression vectors is currently in use.
References: Dröge-Laser et als COPS 2018, Weiste et al. 2017; Wehner et al. 2011a, b; Zhang 2015; Weiste et al., 2007
Co-worker: Christoph Weiste, Markus Henninger
Topic C: Pflant-Pathogen-Interaction
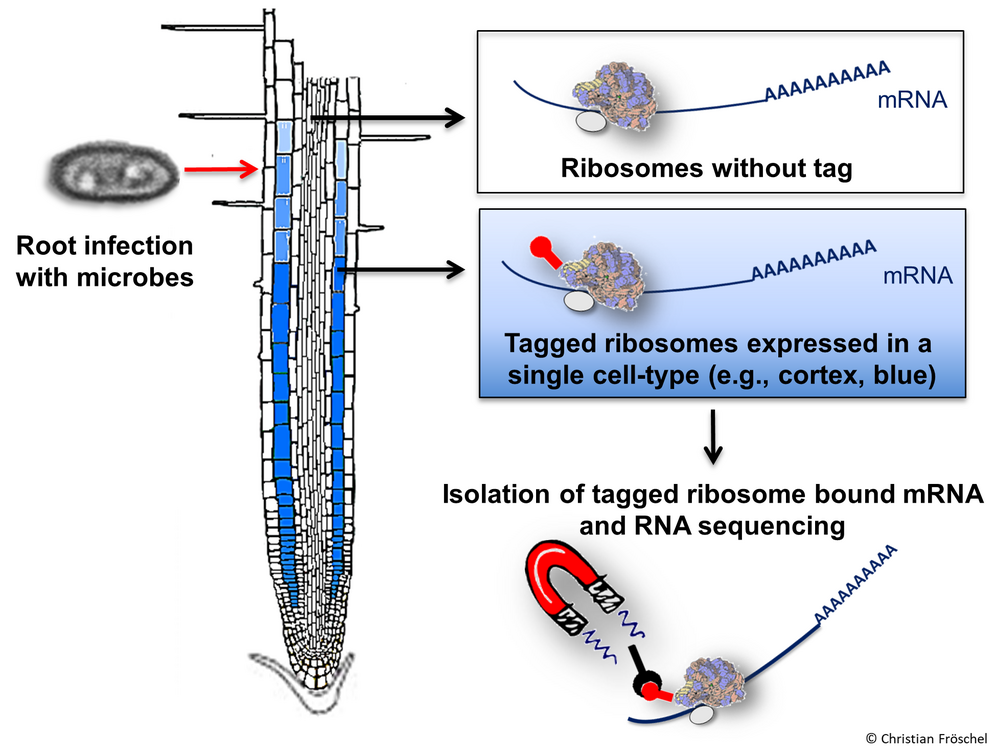
Genome-wide analysis of cell layer-specific expression of pathogen-induced genes in the Arabidopsis root
Although plant roots are in close contact with a variety of microorganisms, inducible root-specific defense responses against pathogens have been poorly characterized. As the root is built by concentric cell-layers, it is anticipated that these layers respond specifically with genetically defined programs. In this project, we established an iTRAPseq methodology (infection-based Translating Ribosome Affinity Purification followed by RNA sequencing) for the model plant Arabidopsis to isolate and analyze cell layer-specific mRNA. Due to cell-type specific expression of FLAG-tagged ribosomal proteins, ribosome/RNA complexes can be co-immunoprecipitated to obtain cell-layer specific expression profiles. We demonstrated that root cell layers respond to microbial infections with specific, genetically defined programs (Fröschel et al., 2021). Cell layer-specific responses to infections with pathogenic and mutualistic fungi are compared further to investigate their relevance for root-microbe interactions.
Literature: Fröschel et al., Cell Host and Microbe 2021
Co-worker: Alexander Marsell
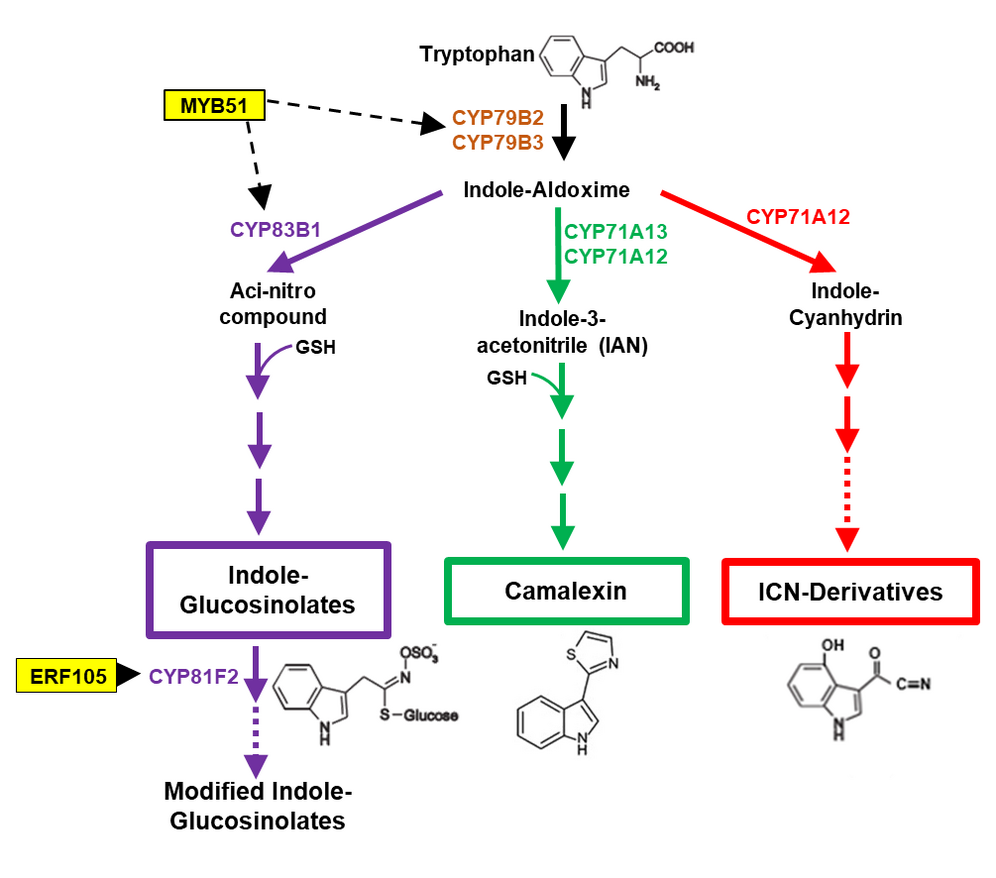
Transcription factor network controlling biosynthesis of tryptophan-derived antimicrobial compounds
As an efficient defense strategy, plants produce secondary metabolites with antimicrobial properties to defeat invading microorganisms. In the model plant Arabidopsis, the antimicrobial compounds camalexin, indole glucosinolates (IG), and ICN derivatives are derived from the amino acid tryptophan (Trp). The complex Trp-dependent secondary metabolism is regulated in a very coordinated manner at the transcriptional level and, thus, represents an ideal model system for the analysis of regulatory transcriptional networks. Using "screening" approaches, we were able to identify key regulators of the IG branch, such as some members of the ERF (Ethylene Response Factor) and MYB (MYB domain protein) transcription factor families. The goal of this project is to identify additional "master" regulators in the Trp-dependent secondary metabolism.
References: Iven et al., Mol. Plant 2012; Fröschel et al., MPMI 2019; Fröschel et al., Cell Host and Microbe 2021
Co-worker:
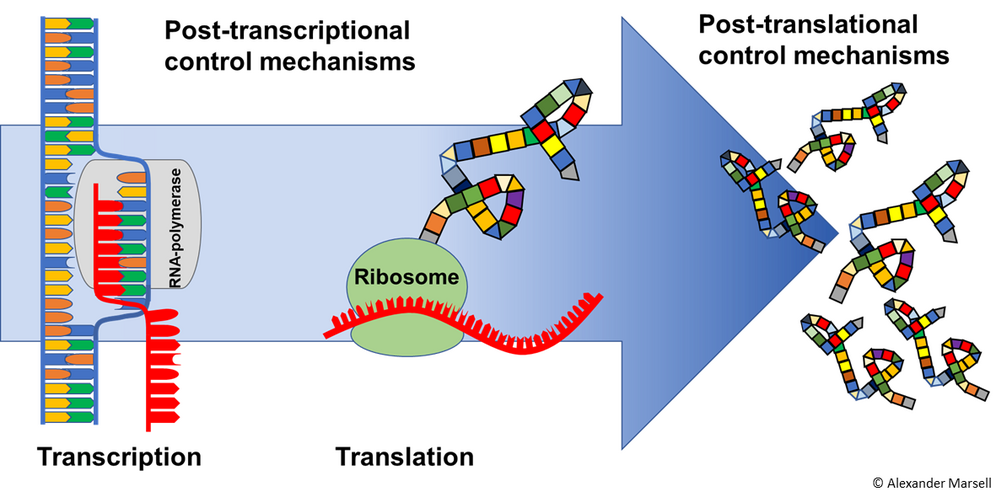
Translational control of pathogen-regulated gene expression in the Arabidopsis root: genome-wide and gene-specific approaches
Recent studies have shown that translational reprogramming is a rapid mechanism to respond to pathogens. In a dynamic environment, translational control of gene expression contributes significantly to plant survival by fine-tuning protein synthesis under optimal or energy-limiting conditions. Using the TRAPseq technique (Translating Ribosome Affinity Purification followed by RNA sequencing), we have the ability to determine the translatome by immunoprecipitation of labelled ribosome/mRNA complexes. Comparison of translatome and transcriptome shows little correlation after pathogen infection of the root. Studies after infection with leaf pathogens led to similar results (Xu et al., 2017), highlighting the importance of translational control after pathogen infection. The goal of our project is to decipher mechanisms that adjust translation in Arabidopsis roots in response to the pathogen Verticillium longisporum.
Literature: Fröschel et al., Cell Host and Microbe 2021
Researcher: Alexander Marsell