Microbial Rhodopsins
Microbial Rhodopsins: Channel Rhodopsins and Pump Rhodopsins
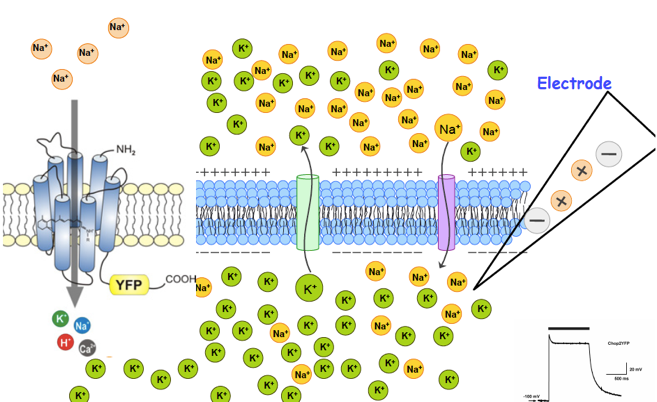
Channelrhodopsins are membrane channels, directly gated by light, which we identified for the first time in 2002 as cation channels with high proton conductivity. Naturally they occur in the membrane of microbes (so far discovered in single- and multi-cellular algae). Their characterization succeeded by heterologous expression in oocytes of the clawed toad Xenopus laevis. In 2003, we described for the first time the properties of channelrhodopsin-2 (ChR2), a directly light-gated cation channel from the unicellular green alga Chlamydomonas reinhardtii. We showed relatively large light-activated inward currents, carried by H+, Na+, and even Ca2+, at negative membrane potential in oocytes and human cultured (HEK293) and other cells, and suggested its use as a "tool" in research. The use of ChR2 in the neurosciences (triggering action potentials by light) was soon called optogenetics and led to a novel experimental approach, which is becoming increasingly widespread. Although we already described the expression of the long-known, light-activated proton pump bacteriorhodopsin (BR) from the archaeum Halobacterium salinarum in oocytes as early as 1995 and showed light-induced outward currents, its use as an optogenetic tool was considered only after the successful use of ChR2. Even before our expression in animal cells, it was known that BR is a light-driven proton pump, i.e. BR absorbs a photon and can use a (small) part of this energy for the active transport of a H+ ion to the outside of the cell. This transport is called active because it takes place even against an electrochemical potential, i.e. against the membrane potential and/or a H+ ion concentration gradient. In contrast, the ion transport of the channel rhodopsins is passive, i.e. the transport direction is determined by the electrochemical potential. The first actively pumping rhodopsin, which was used as an optogenetic tool, following our recommendation and in a collaboration, is halorhodopsin (HR), a light-driven chloride pump. HR pumps chloride into the cell, thus leading to a light-induced hyperpolarization. Therefore, HR can be used for the light-induced prevention of action potential firing (Zhang et al., 2007, Nature). In the course of the last years since our discovery and application, more and more light-controlled channels and pumps have been discovered in nature and their advantages in the application in optogenetics have been described. The most interesting seem the newly discovered natural anion channelrhodopsins (ACR) from sea algae. The cation permeable channelrhodopsins have recently been modified by mutagenesis in their expression strength, kinetics, absorption maxima and ion specificity, examples from our group are: Dawydow et al., 2014; Scholz et al., 2017; Duan et al., 2019.