Bone morphogenetic proteins
Bone morphogenetic proteins - BMPs as key regulators of bone and tissue homeostasis
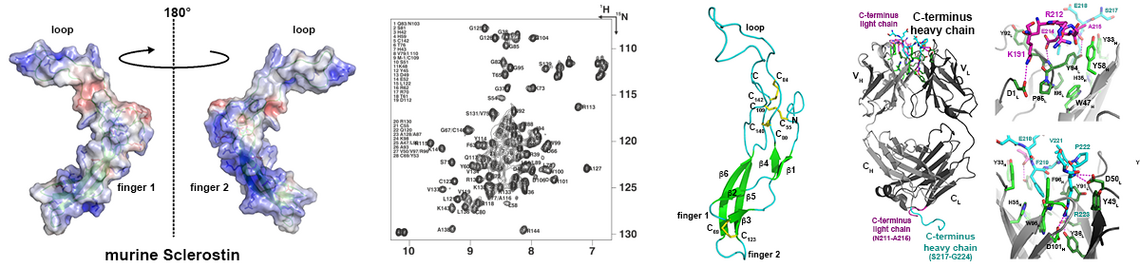
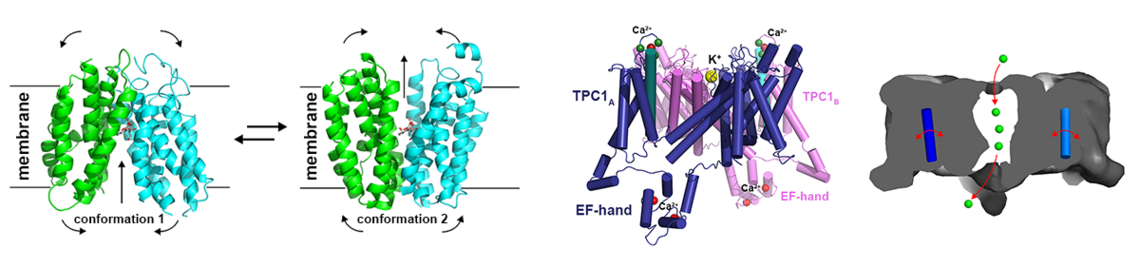
Bone morphogenetic proteins (BMPs) - isolated for the first time 1965 by Marshall Urist from bone - mark the beginning of modern regenerative medicine, today also associated with the term tissue engineering. As suggested from their designation implantation of these proteins in muscle tissue results in the formation of ectopic bone. Hence these proteins are used today in several therapeutic applications, e.g. for the treatment of so-called non-union fractures (e.g. long bone fractures, which hold a defect above a critical size preventing self-healing of the fracture) or for spinal fusions to treat lumbar disc degeneration. Meanwhile more than 30 members of the Transforming Growth Factor (TGF)β family are known, with BMPs forming the largest subgroup. The growth factors activate their receptors by assembling two different types of transmembrane serine/threonine kinase receptors, termed type I and type II, into a heteromeric receptor complex. This assembling subsequently leads to activation of various cytoplasmic signaling cascades such as the SMAD or MAP kinase pathways.
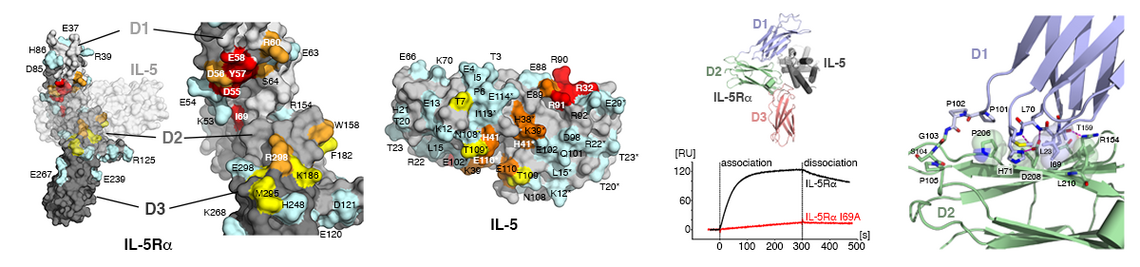
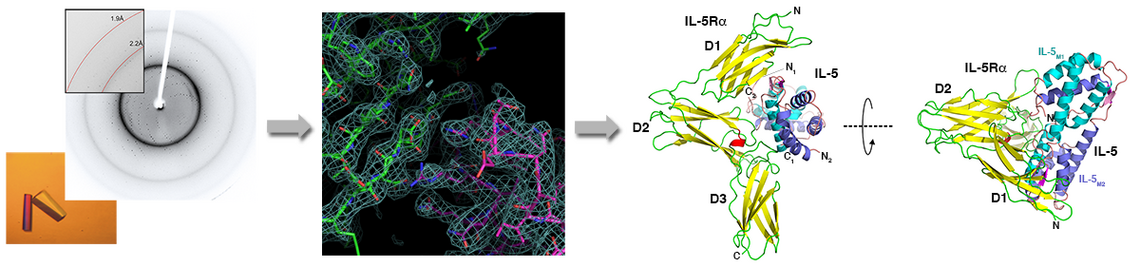
Remarkably, the rather large number of TGFb factors faces only a limited set of 12 receptors, five type I and seven type II receptors. This numeral discrepancy strongly contrasts the plethora of functions encoded by these proteins. Consequently a particular receptor usually binds more than one growth factor and, in addition, many of these factors bind more than one receptor of either subgroup. This firstly raises the question whether and how TGFbs/BMPs transduce ligand-specific signals in spite of this massive ligand-receptor promiscuity leading to seemingly identical receptor assemblies by different TGFb members. Secondly, how are the epitopes in the ligands and receptors formed to allow for such specific/unspecific protein-protein interactions.
From our various structure analyses we could unravel the mechanisms underlying the promiscuous interaction between TGFβ ligands and receptors (e.g. 1-3; for recent reviews: 4-6). The knowledge enables us to design and produce TGFb/BMP factors with defined receptor binding properties. Since the receptors are often expressed in a cell-type specific manner, such designer BMPs can exert defined functionalities. Such novel BMPs present interesting and highly useful tools for new therapeutic interventions, e.g. for the treatment of degenerative bone and cartilage diseases.
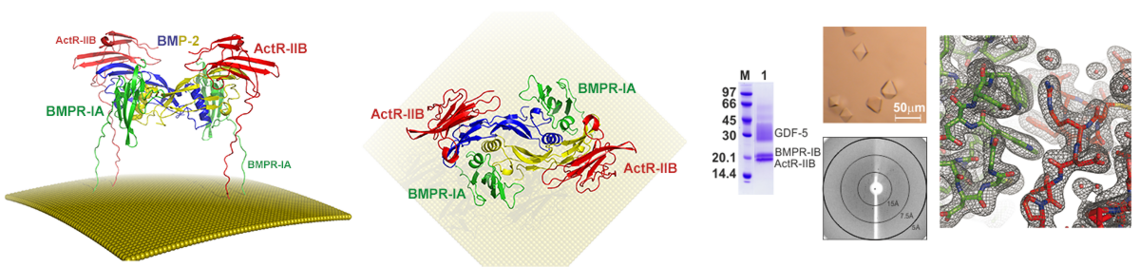
- Klammert U, Mueller TD, Hellmann TV, Wuerzler KK, Kotzsch A, Schliermann A, Schmitz W, Kuebler AC, Sebald W, Nickel J: GDF-5 can act as a context-dependent BMP-2 antagonist. BMC Biol 2015, 13:77.
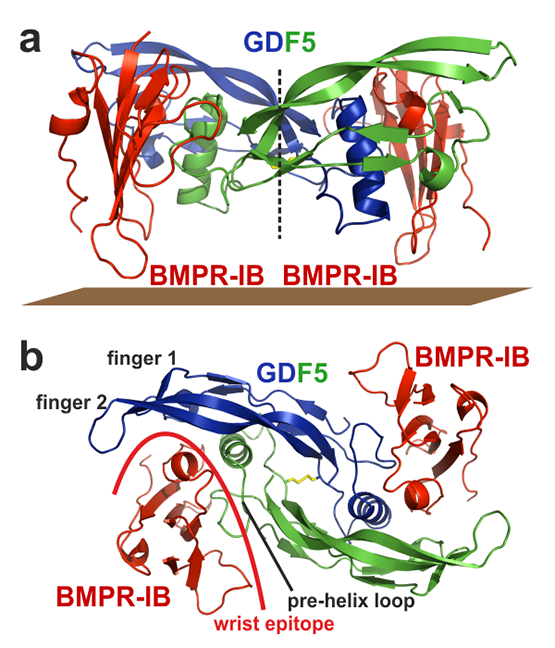
- Kotzsch A, Nickel J, Seher A, Sebald W, Muller TD: Crystal structure analysis reveals a spring-loaded latch as molecular mechanism for GDF-5-type I receptor specificity. EMBO J 2009, 28(7):937-947.
- Kotzsch A, Nickel J, Seher A, Heinecke K, van Geersdaele L, Herrmann T, Sebald W, Mueller TD: Structure analysis of bone morphogenetic protein-2 type I receptor complexes reveals a mechanism of receptor inactivation in juvenile polyposis syndrome. J Biol Chem 2008, 283(9):5876-5887.
- Yadin D, Knaus P, Mueller TD: Structural insights into BMP receptors: Specificity, activation and inhibition. Cytokine Growth Factor Rev 2016, 27:13-34.
- Hinck AP, Mueller TD, Springer TA: Structural Biology and Evolution of the TGF-beta Family. Cold Spring Harb Perspect Biol 2016, 8(12).
- Mueller TD: Mechanisms of BMP-Receptor Interaction and Activation. Vitam Horm 2015, 99:1-61.