The Chlamydia intracellular niche
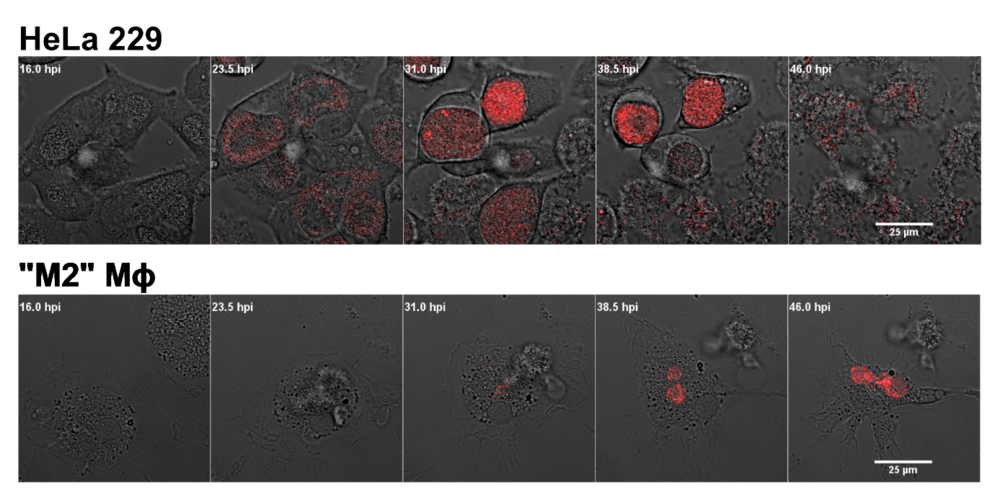
Chlamydia trachomatis can readily infect epithelial cells lining the genital tract. Since professional phagocytic cells, such as monocytes and macrophages, can be present within this niche, these cell types are also likely to be infected. Both macrophages and monocytes can, in fact, be used as Trojan horses, for bacterial dissemination to the lymphatic system.
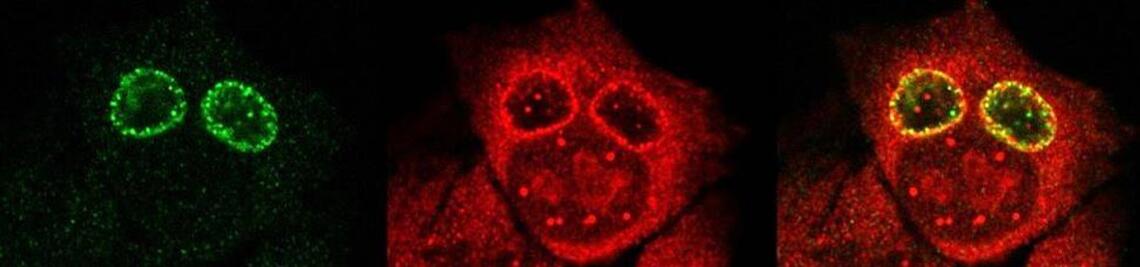
The intracellular fate of Chlamydia in macrophages, is drastically different from the classical developmental cycle observed in epithelial cells. Recent work suggests that alternatively activated macrophages (“M2”), unlike classically activated macrophages (“M1”), are a suitable replicative niche for Chlamydia.
We aim at a better understanding of the dynamics of chlamydial infection within different host cell types and the metabolic and transcriptomic signatures that govern it.
By using different host cell systems, cultured and primary, both non-professional (e.g., epithelial cells) and professional phagocytes (e.g., macrophages) of human origin, we aim at a better understanding of the metabolic alterations of Chlamydia in different intracellular states (i.e. in proliferating RBs and non-replicative, but infectious EBs) as well as the transcriptomic patterns of both host and pathogens. We hope to shed light on the metabolic reprogramming of primary host cells upon interaction with an obligate intracellular bacterial pathogen.