Collaborative Research
CRC 1583: DECIDE (DECisions in Infectious DisEases)
Infectious diseases are a major cause of suffering, morbidity and mortality worldwide. In particular, the worldwide increase of multidrug-resistant pathogens and the constant emergence of new human pathogens pose immense challenges to modern medicine.
Pathogen-directed therapies often lead to successful treatment of the patient. However, antibiotic, antifungal or antiviral drugs cause the emergence of resistant germs. Moreover, current therapeutic approaches do not take into account the host immune response, which often contributes to the fatal outcome of many infectious diseases. Therefore, combating the pathogen alone is not always sufficient.
Re-thinking therapeutic options is therefore necessary. Treatment strategies that focus on optimizing the host response rather than eradicating the pathogen itself are urgently needed. In particular, processes that transform an initially harmless infection into a severe or chronic disease could be exploited for future therapeutic interventions, but have been little explored up to now.
The Collaborative Research Centre DECIDE (DECisions in Infectious DisEases) aims to identify molecular mechanisms within the host that control the course of infectious diseases. In particular, three key decisions that determine the clinical outcome and severity of infections are being investigated:
(1) containment versus active infection after initial contact
(2) active/acute versus persistant/chronic infection
(3) localized infection versus systemic spreading
DECIDE researchers' extensive experience with a repertoire of pathogens allows them to study the interactions between microbes, host barrier tissues, and the immune system, as well as the microbiota in a unique multi-layered systems approach. In this way, we aim to identify overarching common and pathogen-specific molecular decision points in infectious processes that can serve as the basis for new prevention and treatment approaches for infectious diseases.
Inno4Vac - innovations to accelerate vaccine development and manufacture
Inno4Vac is a new interdisciplinary project funded by the Innovative Medicines Initiative 2 (IMI2) that aims to foster health innovation by incorporating scientific and technological breakthrough from the academic and biotech sectors into industry. It is coordinated by the European Vaccine Initiative (Germany), with the support from the Sclavo Vaccines Association (Italy), for the scientific coordination, and involves 41 partners from 11 different European countries, including 37 academic institutions and SMEs, as well as GSK, Sanofi Pasteur, CureVac and Takeda as industry partners.
Within this Inno4Vac initiative the Chair of Microbiology aims at the development of complex infection models of the urovaginal mucosa with features close to the native human tissue regarding architecture and physiology as well as cell autonomous immune response. These models are used to explore the feasibility of exploiting immune cells of innate and adaptive immunity. Our focus is on Neisseria gonorrhoeae to establish infection models. These tissue models will be used for testing and validating vaccination strategies.
GRK 2581: Metabolism, topology and compartmentalization of membrane proximal lipid and signaling components in infection
Targeted intervention of sphingolipid turnover has proven to be a successful strategy in inflammation, but its potential as a target in controlling infectious diseases at the level of metabolism and immune controls requires further definition. Therefore, the GRK 2581 aims to identify and validate targets for novel anti-infective or immunotherapeutic strategies targeting infectious diseases at the level of modulation of the sphingolipid metabolism.
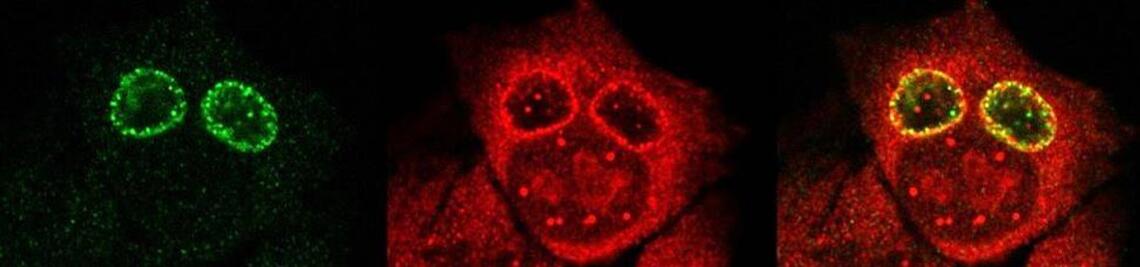
GRK 2243: Understanding Ubiquitylation: From Molecular Mechanisms to Disease
The research focus of the research training group GRK 2243 is the elucidation of biochemical and pathogenic mechanisms underlying diseases that are linked to the ubiquitin system. The research program is aimed at generating a thorough mechanistic understanding of disease-relevant enzymes of the ubiquitin system, which will guide the subsequent molecular and cellular analysis of the pathogenic consequences of their malfunction.