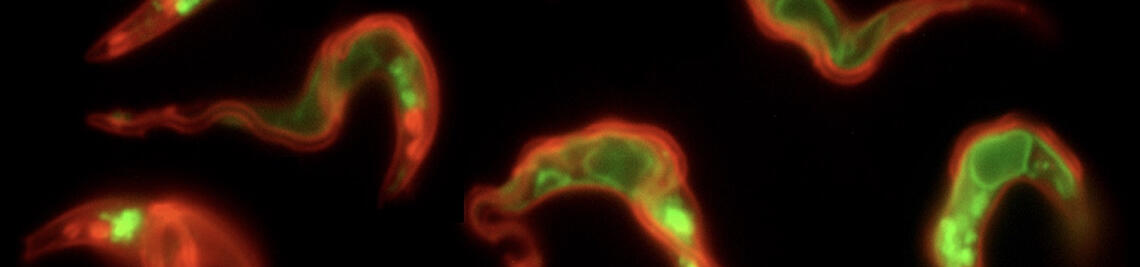
PARASITE MOTILITY
Parasite Motility (Engstler lab)
The trypanosome flagellum always beats.
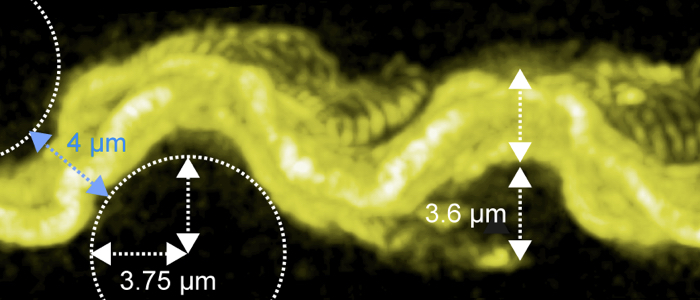
Motion is essential for immune escape and tsetse passage - and it is a very complex business.
How do trypanosomes swim? It's much more complex than thought
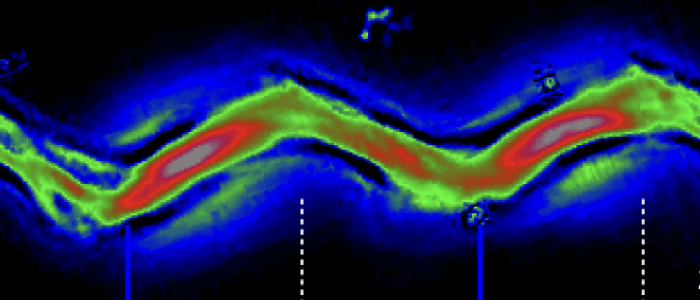
We found that the trypanosome flagellum can productively beat from both ends. This is unique in nature and essential for parasite virulence.
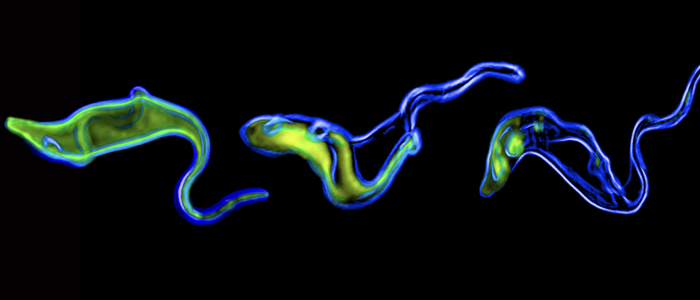
Trypanosomes also swim in the tsetse fly

The trypanosome's journey through the tsetse fly takes several weeks.
Microswimmers
Studies on the motility of microorganisms have focused on relatively few model organisms, including the bacterium E. coli, eukaryotic spermatozoa or the small green algae Chlamydomonas. The seminal work on bacterial motion in particular has been fundamental to teaching us about the swimming of single cells at low Reynolds numbers. More recently the formation of spatiotemporal patterns, e.g. the self-organisation of microswimmers into dynamic vortices and in the vicinity of planar surfaces, has been reported. Large-scale coordination of cells may well be mediated by hydrodynamic interactions that do not depend on the exchange of chemical signals.
Highly adpated

As an adaptation to the varying microenvironments within the mammalian host, trypanosomes modulate the beat direction of their flagellum in response to purely biomechanical cues. The host microenvironment thus determines in which direction the trypanosome flagellum beats, and hence in which direction the cell moves. In the human bloodstream, the deadly parasites are precisely biomechanically adapted to the presence of blood cells and exhibit very rapid forward movement, which is essential for clearing host antibodies from the cell surface. Under more rigorous confinement the flagellar beat reverses and the cells swim backwards, thereby avoiding getting trapped (e.g. in tissue spaces). In the absence of any confinement, beat reversal occurs frequently and randomly, which retards motion. The concept of pathogens biomechanically exploiting the host microenvironment for efficient motility and hence for persistence is interesting, as is the triggering of flagellar beat reversal under confinement.
Trypanosomes also swim in the tsetse fly. We found that depending on their location and developmental stage, the unicellular parasites move either singly or in huge swarms. They display collective behaviour and can congregate or navigate in strong confinement. The tsetse-trypanosome system is thus ideally suited for a systems biophysical exploration of microbial motion.
Select publications
Schuster, S., Krüger, T., Subota, I., Thusek, S., Rotureau, B., Beilhack, A., Engstler, M. (2017) Developmental adaptations of trypanosome motility to the tsetse fly host environments unravel a multifaceted in vivo microswimmer system. eLife, 2017 Aug 15;6. pii: e27656. doi: 10.7554/eLife.27656. PubMed PMID: 28807106; PubMedCentral PMCID: PMC5570225.
Glogger, M; Stichler, S; Subota, I; Bertlein, S; Spindler, M; Tessmar, J; Groll, J; Engstler, M; Fenz, SF (2017) Live-cell super-resolution imaging of intrinsically fast moving flagellates, Journal of Physics D: Applied Physics,50,7,074004
Bargul JL, Jung J, McOdimba FA, Omogo CO, Adung'a VO, Krüger T, Masiga DK, Engstler M. (2016) Species-Specific Adaptations of Trypanosome Morphology and Motility to the Mammalian Host. PLoS Pathog. 2016 Feb 12;12(2):e1005448. doi: 10.1371/journal.ppat.1005448. ePMID: 26871910
Krüger T, Engstler M. (2015) Flagellar motility in eukaryotic human parasites. Semin Cell Dev Biol. 2015 Oct;46:113-27. doi: 10.1016/j.semcdb.2015.10.034. PMID: 26523344
Alizadehrad D, Krüger T, Engstler M, Stark H (2015) Simulating the complex cell design of Trypanosoma brucei and its motility. PLoS Comput Biol 11: e1003967.
Heddergott N, Krüger T, Babu SB, Wei A, Stellamanns E, Uppaluri S, Pfohl T, Stark H, Engstler M (2012) Trypanosome motion represents an adaptation to the crowded environment of the vertebrate bloodstream. PLoS Pathog 8: e1003023.
Uppaluri S, Nagler J, Stellamanns E, Heddergott N, Herminghaus S, Engstler M, Pfohl T (2011) Impact of microscopic motility on the swimming behavior of parasites: straighter trypanosomes are more directional. PLoS Comput Biol 7: e1002058.
Engstler M, Pfohl T, Herminghaus S, Boshart M, Wiegertjes G, Heddergott N, Overath P (2007) Hydrodynamic flow-mediated protein sorting on the cell surface of trypanosomes. Cell 131: 505–515.